Hyamax® Mesotherapy COGN 3, Skin Perfect Medical Aesthetics Manufacture, Support Wholesale and Custom
- Model
- Mesotherapy COGN 3
Item specifics
- Concentration
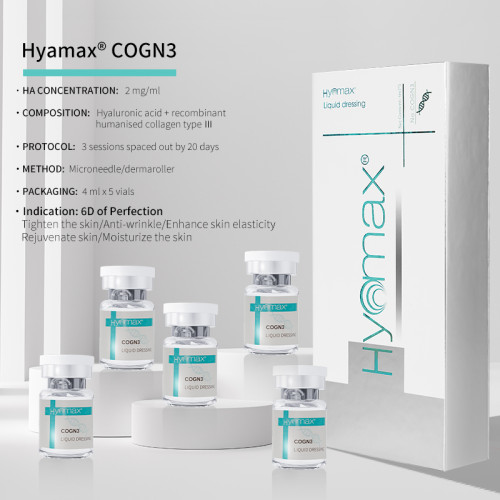
- 2 mg/ml
- Specification
- 4 ml x 5 vials
Review
Description

Hyamax® Mesotherapy COGN 3
Hyamax® Mesotherapy COGN 3 is hyaluronic acid plus recombinant human collagen and 2 amino acid to tighten, anti-wrinkle, boost elasticity and rejuvenate your skin.
Ageing is one of the main causes of collagen loss. The body's metabolism slows down, collagen is in short supply and the skin lacks support and wrinkles. Dry conditions, or prolonged UV exposure, also tend to lead to poorly rounded skin tissue and collagen loss.
Recombinant collagen, also known as COGN, is a collagen prepared using DNA recombination technology. Genetic sequence of the recombinant collagen is highly compatible with the human gene sequence, making it more histocompatible and can be directly absorbed by the body and participate in the construction of collagen to help with tissue repair and site filling.
Hyamax® Mesotherapy COGN is hyaluronic acid plus recombinant human collagen and 2 amino acid. It can tighten the skin, fight wrinkles, increase skin elasticity, rejuvenate the skin and moisturise.

Name | Hyamax® Mesotherapy COGN3 | ||
Attribute | Non-Crosslinked Hyaluronic Acid | ||
Method | Microneedle / Dermaroller | ||
Concentration | 2mg/ml | ||
Injection Depth | Superfacial Dermis | ||
Effect Duration | 3 Months | ||
Treatment | 3 Sessions spaced out by 20 days | ||
Storage Condition | ·For maximum stability and shelf life, the temperature range for storage is 2°C to 25°C, optimally 10°C, and avoid freezing and direct sunlight. ·Expiry date is indicated on each individual package. | ||
Indications for use | Moisturizing Anti-wrinkle Tightening Repair Elasticity Plumpness | ||